-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
Beyond the Scale: GLP-1 Receptor Agonists and the New Frontier of Cardio-Renal-Metabolic Medicine
A Drug Class Reimagined
When glucagon-like peptide-1 (GLP-1) receptor agonists first entered clinical practice, they were celebrated primarily as tools for glycemic control and, later, for their striking capacity to reduce body weight. That framing, however compelling, has always sold the molecule short. A growing and increasingly convergent body of evidence now reveals that GLP-1 receptor agonists operate at a far more fundamental biological level — one that extends well beyond adipose tissue and the bathroom scale. They are, in the most precise sense, upstream metabolic modulators: agents that intervene at the root of systemic metabolic dysfunction and, in doing so, simultaneously protect the heart, the kidneys, and the airway from the cumulative ravages of that dysfunction.
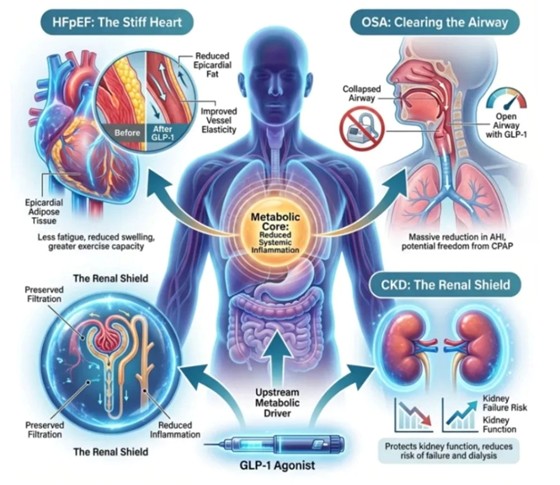
To appreciate the full scope of what these medications can offer, one must first understand the concept of the metabolic core — and then trace how disturbances in that core propagate outward to damage organ systems that, on the surface, appear unrelated. The figure above offers precisely that synthesis, illustrating how a single pharmacological intervention can reshape the physiological trajectory of three of the most prevalent and morbid conditions in modern medicine: heart failure with preserved ejection fraction (HFpEF), obstructive sleep apnea (OSA), and chronic kidney disease (CKD).
To appreciate the full scope of what these medications can offer, one must first understand the concept of the metabolic core — and then trace how disturbances in that core propagate outward to damage organ systems that, on the surface, appear unrelated. The figure above offers precisely that synthesis, illustrating how a single pharmacological intervention can reshape the physiological trajectory of three of the most prevalent and morbid conditions in modern medicine: heart failure with preserved ejection fraction (HFpEF), obstructive sleep apnea (OSA), and chronic kidney disease (CKD).
The Metabolic Core: Targeting the Upstream Driver
The unifying pathophysiological thread connecting HFpEF, OSA, and CKD is not merely obesity or hyperglycemia — it is chronic, low-grade systemic inflammation. This inflammatory state, perpetuated by visceral adiposity, insulin resistance, and metabolic overload, acts as the biological accelerant that stiffens myocardial tissue, narrows upper airways, and scars glomerular capillaries. Treating each of these sequelae in isolation, as medicine has long done, is the clinical equivalent of mopping the floor without turning off the tap.
GLP-1 receptor agonists turn off the tap. By engaging the GLP-1 receptor across multiple tissue types — including the central nervous system, the gastrointestinal tract, the pancreas, the heart, and the vasculature — these agents orchestrate a coordinated reduction in systemic inflammatory burden. This is not a peripheral effect of weight loss, though weight loss clearly amplifies the benefit. Even in studies controlling for changes in body mass, GLP-1 agonists demonstrate direct anti-inflammatory activity through suppression of pro-inflammatory cytokines, attenuation of oxidative stress, and modulation of macrophage polarization within adipose and organ tissue. It is this upstream action that unlocks the cascade of downstream benefits illustrated in Figure 1.
Understanding the metabolic core as the therapeutic target reframes our thinking about what GLP-1 agonists are actually treating. They are not simply diabetes medications repurposed for weight loss. They are systemic anti-inflammatory agents that happen to manifest their most visible effects on the scale — while quietly and profoundly reshaping the physiology of the organs that determine survival and quality of life.
GLP-1 receptor agonists turn off the tap. By engaging the GLP-1 receptor across multiple tissue types — including the central nervous system, the gastrointestinal tract, the pancreas, the heart, and the vasculature — these agents orchestrate a coordinated reduction in systemic inflammatory burden. This is not a peripheral effect of weight loss, though weight loss clearly amplifies the benefit. Even in studies controlling for changes in body mass, GLP-1 agonists demonstrate direct anti-inflammatory activity through suppression of pro-inflammatory cytokines, attenuation of oxidative stress, and modulation of macrophage polarization within adipose and organ tissue. It is this upstream action that unlocks the cascade of downstream benefits illustrated in Figure 1.
Understanding the metabolic core as the therapeutic target reframes our thinking about what GLP-1 agonists are actually treating. They are not simply diabetes medications repurposed for weight loss. They are systemic anti-inflammatory agents that happen to manifest their most visible effects on the scale — while quietly and profoundly reshaping the physiology of the organs that determine survival and quality of life.
The Stiff Heart: GLP-1 Agonists in Heart Failure with Preserved Ejection Fraction
The Problem of Diastolic Dysfunction
Heart failure with preserved ejection fraction represents one of the most therapeutically challenging syndromes in contemporary cardiology. Unlike its counterpart with reduced ejection fraction, HFpEF is characterized not by a weakened pump, but by a stiff one — a ventricle that contracts normally but fails to relax, filling poorly and transmitting elevated pressures back into the pulmonary circulation. Patients present with exertional dyspnea, fatigue, and peripheral edema, yet their echocardiographic systolic function appears intact. This disconnect has long frustrated clinicians and eluded conventional heart failure therapies.
The structural substrate of HFpEF is now well understood. Epicardial adipose tissue — the visceral fat depot that drapes directly over the myocardium and pericardium — is not a passive bystander. It is a metabolically active organ that secretes adipokines, inflammatory cytokines, and reactive oxygen species directly into the myocardial interstitium. This local lipotoxic and inflammatory environment drives myocardial fibrosis, impairs cardiomyocyte energetics, and stiffens the ventricular wall. The result is the diastolic dysfunction that defines HFpEF.
The GLP-1 Effect on the Myocardium
GLP-1 receptor agonists address the structural pathology of HFpEF through two complementary and synergistic mechanisms. First, by reducing overall visceral adiposity, they diminish the volume of epicardial fat that bathes the myocardium in inflammatory signals — a change that can be quantified on cardiac MRI and that correlates directly with improvements in diastolic parameters. Second, and perhaps more fundamentally, they improve vascular elasticity throughout the coronary and systemic vasculature, reducing afterload and the pulsatile hemodynamic burden that the stiff ventricle is least equipped to handle.
The clinical dividend of these structural changes is meaningful and patient-centered. Trials such as STEP-HFpEF have demonstrated that GLP-1 agonist therapy in patients with obesity-related HFpEF significantly reduces symptom burden, as measured by validated quality-of-life instruments, and improves functional capacity on six-minute walk testing. Patients report less fatigue, a reduction in lower extremity swelling, and a demonstrable improvement in their ability to perform activities of daily living. These are not surrogate endpoints — they are the outcomes that patients and their families care about most.
For a condition that has historically offered patients little more than symptom management with diuretics, the emergence of GLP-1 agonists as disease-modifying therapy in HFpEF represents a genuine paradigm shift. The stiff heart, it turns out, can be coaxed toward greater compliance — not through a cardiac medication at all, but through an agent that addresses the metabolic milieu in which the heart is forced to operate.
Heart failure with preserved ejection fraction represents one of the most therapeutically challenging syndromes in contemporary cardiology. Unlike its counterpart with reduced ejection fraction, HFpEF is characterized not by a weakened pump, but by a stiff one — a ventricle that contracts normally but fails to relax, filling poorly and transmitting elevated pressures back into the pulmonary circulation. Patients present with exertional dyspnea, fatigue, and peripheral edema, yet their echocardiographic systolic function appears intact. This disconnect has long frustrated clinicians and eluded conventional heart failure therapies.
The structural substrate of HFpEF is now well understood. Epicardial adipose tissue — the visceral fat depot that drapes directly over the myocardium and pericardium — is not a passive bystander. It is a metabolically active organ that secretes adipokines, inflammatory cytokines, and reactive oxygen species directly into the myocardial interstitium. This local lipotoxic and inflammatory environment drives myocardial fibrosis, impairs cardiomyocyte energetics, and stiffens the ventricular wall. The result is the diastolic dysfunction that defines HFpEF.
The GLP-1 Effect on the Myocardium
GLP-1 receptor agonists address the structural pathology of HFpEF through two complementary and synergistic mechanisms. First, by reducing overall visceral adiposity, they diminish the volume of epicardial fat that bathes the myocardium in inflammatory signals — a change that can be quantified on cardiac MRI and that correlates directly with improvements in diastolic parameters. Second, and perhaps more fundamentally, they improve vascular elasticity throughout the coronary and systemic vasculature, reducing afterload and the pulsatile hemodynamic burden that the stiff ventricle is least equipped to handle.
The clinical dividend of these structural changes is meaningful and patient-centered. Trials such as STEP-HFpEF have demonstrated that GLP-1 agonist therapy in patients with obesity-related HFpEF significantly reduces symptom burden, as measured by validated quality-of-life instruments, and improves functional capacity on six-minute walk testing. Patients report less fatigue, a reduction in lower extremity swelling, and a demonstrable improvement in their ability to perform activities of daily living. These are not surrogate endpoints — they are the outcomes that patients and their families care about most.
For a condition that has historically offered patients little more than symptom management with diuretics, the emergence of GLP-1 agonists as disease-modifying therapy in HFpEF represents a genuine paradigm shift. The stiff heart, it turns out, can be coaxed toward greater compliance — not through a cardiac medication at all, but through an agent that addresses the metabolic milieu in which the heart is forced to operate.
Clearing the Airway: GLP-1 Agonists and Obstructive Sleep Apnea
The Mechanical and Metabolic Roots of OSA
Obstructive sleep apnea is, at its core, a disease of anatomical crowding. When pharyngeal fat deposition and reduced upper airway muscle tone allow the soft tissues of the posterior pharynx to collapse during sleep, airflow ceases, oxygen saturation falls, and the brain is forced to arouse the sleeper — repeatedly, often hundreds of times per night, and rarely to the point of conscious wakefulness. The resulting sleep fragmentation drives daytime somnolence, neurocognitive impairment, sympathetic nervous system activation, and a cardiovascular risk profile that includes hypertension, atrial fibrillation, and increased susceptibility to sudden cardiac death.
Continuous positive airway pressure (CPAP) therapy has long been the standard of care for moderate-to-severe OSA, and it remains highly effective — when tolerated. The uncomfortable truth, however, is that adherence to CPAP is notoriously poor. Roughly half of patients prescribed the device fail to use it for the recommended minimum duration, citing discomfort, claustrophobia, noise, and the practical challenges of travel. For these patients, the apneas continue, the arousals accumulate, and the cardiovascular consequences compound unchecked. An alternative or adjunctive treatment capable of reducing OSA severity without requiring a device worn over the face during sleep would be transformative.
Structural Relief Through Metabolic Intervention
GLP-1 receptor agonists offer precisely that possibility. The SURMOUNT-OSA trial, which evaluated tirzepatide — a dual GLP-1 and GIP receptor agonist — in patients with moderate-to-severe OSA, produced results that surprised even the most optimistic investigators. Participants receiving active treatment experienced a reduction in the apnea-hypopnea index (AHI) that was, in many cases, sufficient to reclassify their disease from severe to mild, and in a notable proportion of patients, to the point where CPAP was no longer clinically indicated.
The mechanism is both mechanical and metabolic. Reduction in upper airway fat deposits directly increases the caliber of the pharyngeal lumen, reducing the propensity for collapse. Simultaneously, reductions in systemic inflammation improve the tone and responsiveness of upper airway dilator muscles — muscles that in metabolically compromised individuals have been shown to be infiltrated with lipid and rendered sluggish. The open airway illustrated in Figure 1 is not merely a metaphor; it is an anatomical reality demonstrated on imaging in patients who have undergone sustained GLP-1 agonist therapy.
Beyond the immediate relief of apneic events, the downstream cardiovascular implications of reducing OSA severity with GLP-1 therapy are potentially enormous. Sustained reductions in nocturnal hypoxemia, sympathetic surges, and inflammatory activation may translate into meaningful long-term reductions in hypertension, arrhythmia burden, and cardiovascular events — outcomes that CPAP alone has struggled to demonstrate in randomized trials, perhaps because of the adherence problem described above. An effective metabolic intervention, taken once weekly as a subcutaneous injection, sidesteps that adherence barrier entirely.
Obstructive sleep apnea is, at its core, a disease of anatomical crowding. When pharyngeal fat deposition and reduced upper airway muscle tone allow the soft tissues of the posterior pharynx to collapse during sleep, airflow ceases, oxygen saturation falls, and the brain is forced to arouse the sleeper — repeatedly, often hundreds of times per night, and rarely to the point of conscious wakefulness. The resulting sleep fragmentation drives daytime somnolence, neurocognitive impairment, sympathetic nervous system activation, and a cardiovascular risk profile that includes hypertension, atrial fibrillation, and increased susceptibility to sudden cardiac death.
Continuous positive airway pressure (CPAP) therapy has long been the standard of care for moderate-to-severe OSA, and it remains highly effective — when tolerated. The uncomfortable truth, however, is that adherence to CPAP is notoriously poor. Roughly half of patients prescribed the device fail to use it for the recommended minimum duration, citing discomfort, claustrophobia, noise, and the practical challenges of travel. For these patients, the apneas continue, the arousals accumulate, and the cardiovascular consequences compound unchecked. An alternative or adjunctive treatment capable of reducing OSA severity without requiring a device worn over the face during sleep would be transformative.
Structural Relief Through Metabolic Intervention
GLP-1 receptor agonists offer precisely that possibility. The SURMOUNT-OSA trial, which evaluated tirzepatide — a dual GLP-1 and GIP receptor agonist — in patients with moderate-to-severe OSA, produced results that surprised even the most optimistic investigators. Participants receiving active treatment experienced a reduction in the apnea-hypopnea index (AHI) that was, in many cases, sufficient to reclassify their disease from severe to mild, and in a notable proportion of patients, to the point where CPAP was no longer clinically indicated.
The mechanism is both mechanical and metabolic. Reduction in upper airway fat deposits directly increases the caliber of the pharyngeal lumen, reducing the propensity for collapse. Simultaneously, reductions in systemic inflammation improve the tone and responsiveness of upper airway dilator muscles — muscles that in metabolically compromised individuals have been shown to be infiltrated with lipid and rendered sluggish. The open airway illustrated in Figure 1 is not merely a metaphor; it is an anatomical reality demonstrated on imaging in patients who have undergone sustained GLP-1 agonist therapy.
Beyond the immediate relief of apneic events, the downstream cardiovascular implications of reducing OSA severity with GLP-1 therapy are potentially enormous. Sustained reductions in nocturnal hypoxemia, sympathetic surges, and inflammatory activation may translate into meaningful long-term reductions in hypertension, arrhythmia burden, and cardiovascular events — outcomes that CPAP alone has struggled to demonstrate in randomized trials, perhaps because of the adherence problem described above. An effective metabolic intervention, taken once weekly as a subcutaneous injection, sidesteps that adherence barrier entirely.
The Renal Shield: GLP-1 Agonists and Chronic Kidney Disease
The Silent Progression of Renal Metabolic Injury
Chronic kidney disease is the quintessential silent disease. It advances over years and decades, destroying nephrons without generating symptoms until the glomerular filtration rate has fallen to a fraction of its normal value. By the time patients are symptomatic, the damage is often irreversible, and the trajectory leads inexorably toward dialysis, transplantation, and the profound personal and societal costs that accompany renal replacement therapy. The identification of interventions capable of genuinely slowing CKD progression — not merely managing its metabolic consequences — represents one of the most important priorities in nephrology.
The pathophysiology of diabetic and metabolic nephropathy is rooted in the same inflammatory and hemodynamic dysregulation that drives HFpEF and OSA. Intraglomerular hypertension, driven by afferent arteriolar dilation in the setting of insulin resistance and obesity, subjects glomerular capillaries to injurious mechanical stretch. Simultaneously, local and systemic inflammatory cytokines infiltrate the mesangium, promote podocyte injury, and accelerate the deposition of extracellular matrix that gradually scleroses the filtering apparatus. Proteinuria — the clinical hallmark of this process — is both a marker of existing damage and a pathological mediator that accelerates further injury.
Nephroprotection Beyond Glucose Control
The nephroprotective effects of GLP-1 receptor agonists have been documented across multiple large-scale cardiovascular outcomes trials and, more recently, in dedicated renal endpoint studies. The FLOW trial, evaluating semaglutide in patients with type 2 diabetes and CKD, demonstrated a significant reduction in the composite of kidney failure, doubling of serum creatinine, and renal death — a landmark result that has secured GLP-1 agonists a place in nephrology guidelines alongside the established renoprotective agents such as RAAS blockade and SGLT2 inhibitors.
The mechanisms underlying this renoprotection are multifactorial. At the level of the nephron, GLP-1 receptor activation reduces oxidative stress and pro-inflammatory signaling within the tubular epithelium and glomerular endothelium, preserving the structural integrity of the filtration barrier. Reductions in systemic blood pressure and intraglomerular hypertension relieve the mechanical burden on the glomerular capillary wall. And weight loss-mediated reductions in visceral adiposity diminish the adipokine-driven inflammatory milieu that accelerates mesangial expansion and podocyte loss.
Clinically, these mechanisms translate into preserved filtration capacity over time, reduced rates of proteinuria progression, and — most critically — a reduction in the incidence of end-stage renal disease. For patients already living with stage 3 or 4 CKD, the prospect of delaying or avoiding dialysis is not merely a statistical abstraction. It is the difference between a life constrained by thrice-weekly machine dependence and one that retains meaningful autonomy. GLP-1 agonists, by attacking the upstream metabolic drivers of renal injury, are capable of offering that difference to millions of patients worldwide.
Chronic kidney disease is the quintessential silent disease. It advances over years and decades, destroying nephrons without generating symptoms until the glomerular filtration rate has fallen to a fraction of its normal value. By the time patients are symptomatic, the damage is often irreversible, and the trajectory leads inexorably toward dialysis, transplantation, and the profound personal and societal costs that accompany renal replacement therapy. The identification of interventions capable of genuinely slowing CKD progression — not merely managing its metabolic consequences — represents one of the most important priorities in nephrology.
The pathophysiology of diabetic and metabolic nephropathy is rooted in the same inflammatory and hemodynamic dysregulation that drives HFpEF and OSA. Intraglomerular hypertension, driven by afferent arteriolar dilation in the setting of insulin resistance and obesity, subjects glomerular capillaries to injurious mechanical stretch. Simultaneously, local and systemic inflammatory cytokines infiltrate the mesangium, promote podocyte injury, and accelerate the deposition of extracellular matrix that gradually scleroses the filtering apparatus. Proteinuria — the clinical hallmark of this process — is both a marker of existing damage and a pathological mediator that accelerates further injury.
Nephroprotection Beyond Glucose Control
The nephroprotective effects of GLP-1 receptor agonists have been documented across multiple large-scale cardiovascular outcomes trials and, more recently, in dedicated renal endpoint studies. The FLOW trial, evaluating semaglutide in patients with type 2 diabetes and CKD, demonstrated a significant reduction in the composite of kidney failure, doubling of serum creatinine, and renal death — a landmark result that has secured GLP-1 agonists a place in nephrology guidelines alongside the established renoprotective agents such as RAAS blockade and SGLT2 inhibitors.
The mechanisms underlying this renoprotection are multifactorial. At the level of the nephron, GLP-1 receptor activation reduces oxidative stress and pro-inflammatory signaling within the tubular epithelium and glomerular endothelium, preserving the structural integrity of the filtration barrier. Reductions in systemic blood pressure and intraglomerular hypertension relieve the mechanical burden on the glomerular capillary wall. And weight loss-mediated reductions in visceral adiposity diminish the adipokine-driven inflammatory milieu that accelerates mesangial expansion and podocyte loss.
Clinically, these mechanisms translate into preserved filtration capacity over time, reduced rates of proteinuria progression, and — most critically — a reduction in the incidence of end-stage renal disease. For patients already living with stage 3 or 4 CKD, the prospect of delaying or avoiding dialysis is not merely a statistical abstraction. It is the difference between a life constrained by thrice-weekly machine dependence and one that retains meaningful autonomy. GLP-1 agonists, by attacking the upstream metabolic drivers of renal injury, are capable of offering that difference to millions of patients worldwide.
Toward a New Therapeutic Philosophy: Treating the Patient, Not the Organ
The convergence of evidence reviewed here demands a fundamental reconsideration of how we conceptualize the treatment of cardio-renal-metabolic disease. The traditional model — a cardiologist managing the stiff heart, a pulmonologist titrating the CPAP machine, a nephrologist monitoring the creatinine — is not wrong, but it is insufficient. It treats the sequelae while leaving the source largely unaddressed. The metabolic core, the upstream driver of all three conditions, remains inflamed and dysregulated while specialists competently manage its end-organ manifestations.
GLP-1 receptor agonists offer a different kind of medicine — one that treats the patient rather than the organ. By targeting the shared biological substrate of HFpEF, OSA, and CKD simultaneously, a single weekly injection has the potential to bend the trajectory of all three conditions at once, with a safety profile that is increasingly well characterized and a tolerability record that continues to improve with next-generation formulations. The practical implications for health systems are as significant as the clinical ones: a patient whose HFpEF, OSA, and CKD are all stabilized by a single metabolic intervention consumes fewer hospitalizations, fewer specialist encounters, and fewer downstream procedural resources.
This is not to suggest that GLP-1 agonists are a universal cure or that organ-specific management becomes obsolete. Patients with advanced CKD still require careful nephrological oversight. Patients with severe HFpEF still benefit from optimized diuresis and tailored hemodynamic management. Patients with persistent OSA after weight loss may still require CPAP. The point is integration, not replacement: GLP-1 agonist therapy as the metabolic foundation upon which organ-specific interventions are layered — a foundation that, by reducing systemic inflammation and visceral adiposity, makes every other intervention more likely to succeed.
GLP-1 receptor agonists offer a different kind of medicine — one that treats the patient rather than the organ. By targeting the shared biological substrate of HFpEF, OSA, and CKD simultaneously, a single weekly injection has the potential to bend the trajectory of all three conditions at once, with a safety profile that is increasingly well characterized and a tolerability record that continues to improve with next-generation formulations. The practical implications for health systems are as significant as the clinical ones: a patient whose HFpEF, OSA, and CKD are all stabilized by a single metabolic intervention consumes fewer hospitalizations, fewer specialist encounters, and fewer downstream procedural resources.
This is not to suggest that GLP-1 agonists are a universal cure or that organ-specific management becomes obsolete. Patients with advanced CKD still require careful nephrological oversight. Patients with severe HFpEF still benefit from optimized diuresis and tailored hemodynamic management. Patients with persistent OSA after weight loss may still require CPAP. The point is integration, not replacement: GLP-1 agonist therapy as the metabolic foundation upon which organ-specific interventions are layered — a foundation that, by reducing systemic inflammation and visceral adiposity, makes every other intervention more likely to succeed.
Conclusion: The Scale Was Never the Destination
The public imagination was captured by GLP-1 agonists as weight loss drugs. That capture was understandable — the efficacy data were dramatic, the cultural moment was ripe, and the visible transformation of body weight provided a narrative that complex mechanistic biology never could. But the weight lost is, in the fullest sense, a visible proxy for an invisible and more profound transformation: the reduction of the chronic inflammatory state that sits at the origin of some of the most debilitating and lethal diseases in the human repertoire.
As the evidence matures and the indications expand, it becomes increasingly clear that GLP-1 receptor agonists are among the most consequential pharmacological developments of the past generation — not because they help people fit into smaller clothes, but because they attack the metabolic root of suffering in the heart, the airway, and the kidney simultaneously. The scale, it turns out, was never the destination. It was only the first landmark on a much longer and more significant journey.
As the evidence matures and the indications expand, it becomes increasingly clear that GLP-1 receptor agonists are among the most consequential pharmacological developments of the past generation — not because they help people fit into smaller clothes, but because they attack the metabolic root of suffering in the heart, the airway, and the kidney simultaneously. The scale, it turns out, was never the destination. It was only the first landmark on a much longer and more significant journey.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis