-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
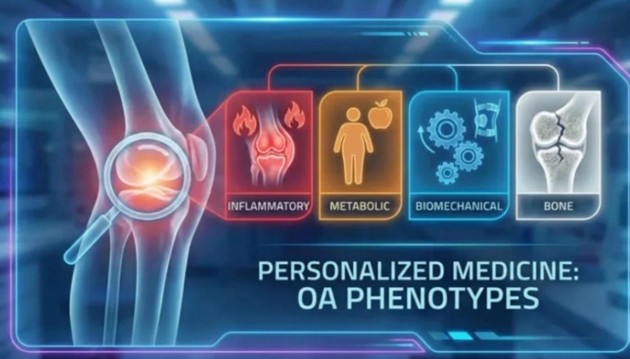
The End of"One-Size-Fits-All": Why Your Osteoarthritis Treatment is Getting Personal
The End of "Wear and Tear": Why One Treatment Can No Longer Fit All
For decades, osteoarthritis was communicated to patients with disarming simplicity: your joints are wearing out. The cartilage is thinning, the bones are rubbing, and there is nothing to be done except manage the pain, lose some weight, and wait for the joint replacement. This narrative, while not entirely wrong, was reductive in a way that had real clinical consequences. It framed OA as a fundamentally passive, inevitable, and biologically uniform process — and it justified a therapeutic approach that was equally undifferentiated. Patients across a vast spectrum of disease mechanisms received the same short-list of interventions: nonsteroidal anti-inflammatory drugs, physiotherapy, corticosteroid injections, and eventual surgical referral. The problem is that these treatments do not work equally well for all patients, and mounting evidence from the past two decades has made clear why: because not all osteoarthritis is biologically the same. Two patients walking into a clinic with identical radiographic findings and pain scores may have arrived there through entirely different pathological pathways — and what resolves inflammation in one will do nothing for the mechanical overloading driving disease in another. The movement toward OA phenotyping is the field's formal acknowledgment of this reality, and it represents the first credible framework for making osteoarthritis management genuinely personalized.
The Inflammatory Phenotype: When the Joint Is on Fire
The inflammatory OA phenotype is characterized by active, localized inflammation within the synovial membrane — the lining of the joint capsule. Patients in this phenotype frequently experience disproportionately severe pain relative to their degree of structural damage, morning stiffness lasting longer than 30 minutes, joint warmth and effusion, and elevated inflammatory markers on laboratory testing. At the cellular level, the synovium of inflammatory-phenotype OA patients shows macrophage infiltration, elevated production of pro-inflammatory cytokines including interleukin-1 beta and tumor necrosis factor alpha, and activation of matrix metalloproteinases that accelerate cartilage degradation. Clinically, these patients respond more robustly to anti-inflammatory interventions — intra-articular corticosteroids, hyaluronic acid injections in appropriate cases, and emerging biologics targeting specific cytokine pathways — than patients whose OA is driven by predominantly structural mechanisms. Identifying this phenotype is therefore not merely academic: it directs the clinician toward treatments with genuine efficacy for the underlying pathology rather than defaulting to analgesics that mask symptoms without modifying disease.
The Metabolic Phenotype: Systemic Disease Written in the Joint
The metabolic OA phenotype is perhaps the most prevalent in modern clinical practice, reflecting the global epidemics of obesity and metabolic syndrome. While it was historically assumed that obesity worsened OA simply by increasing mechanical load on the joint, research has progressively revealed a more complex metabolic contribution: adipose tissue, particularly visceral fat, is a potent endocrine organ that releases pro-inflammatory adipokines — including leptin, adiponectin, and resistin — into systemic circulation, where they directly promote synovial inflammation and chondrocyte apoptosis even in non-weight-bearing joints such as the hands and wrists. Patients with the metabolic phenotype characteristically have OA that is polyarticular rather than confined to a single joint, often in association with dyslipidemia, insulin resistance, or frank type 2 diabetes. The most evidence-based intervention for this phenotype is sustained weight loss — which has been shown not only to reduce mechanical joint loading but to meaningfully lower systemic inflammatory markers and improve patient-reported pain and function scores. Dietary interventions targeting glycemic control and anti-inflammatory eating patterns, combined with structured exercise programs, address the underlying metabolic drivers in a way that pharmacological analgesia alone simply cannot.
The Biomechanical and Bone Phenotypes: Structure, Load, and the Architecture of Failure
The biomechanical OA phenotype encompasses patients in whom structural malalignment, joint instability, muscle weakness, or patterns of mechanical overloading are the primary drivers of cartilage degeneration. Varus or valgus knee deformity, patellofemoral maltracking, ligamentous laxity following injury, occupational kneeling or squatting, and running biomechanics with poor shock absorption all fall within this category. For these patients, the most impactful interventions are those that directly modify joint mechanics: targeted physiotherapy programs to strengthen the quadriceps and hip abductors, orthotic correction of lower limb alignment, gait retraining, and — in cases of significant deformity — surgical correction via osteotomy before joint replacement becomes necessary. The bone phenotype, by contrast, directs attention to the subchondral bone compartment rather than the cartilage surface. Emerging imaging and histological data have identified a distinct subset of OA patients in whom abnormal bone remodeling, subchondral sclerosis, bone marrow lesions, and microfracture formation appear to precede and drive cartilage loss rather than follow it as a secondary consequence. This phenotype may ultimately prove most amenable to bone-targeting agents — including bisphosphonates and anti-resorptive therapies — whose effects on OA outcomes are currently being evaluated in dedicated clinical trials. Together, the biomechanical and bone phenotypes illustrate that the future of OA management lies in treating the architecture of the joint, not just the sensation it produces.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis