-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
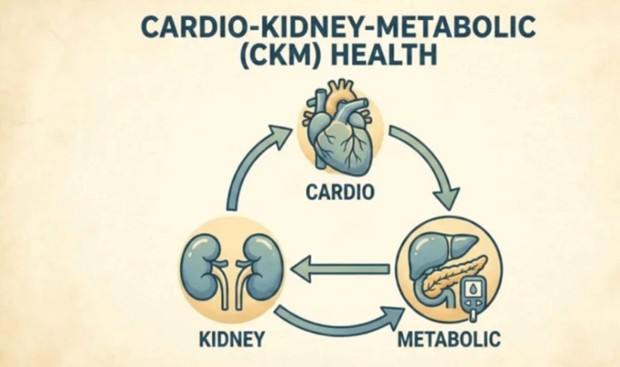
The Silo Is Broken: Why We No Longer Treat Kidneys and Hypertension Separately
The Age of Silos: How Medicine Learned to Divide What the Body Never Separated
For the better part of the twentieth century, the organizing logic of modern medicine was specialization. Cardiologists managed the heart and its vessels; nephrologists managed the kidneys and their filtration function; endocrinologists managed metabolic disorders like diabetes and obesity. Each discipline built its own body of knowledge, its own clinical guidelines, its own pharmacopoeia, and — critically — its own lens through which it interpreted a patient's disease. A person living with type 2 diabetes, hypertension, and proteinuria might be seen by three separate specialists on three separate days, each adjusting one piece of a complex biological puzzle without full visibility of the others. This fragmentation was not born of negligence; it was the natural consequence of the exponential growth of medical knowledge requiring increasingly focused expertise. But as the decades passed and patient outcomes were examined with greater epidemiological rigor, an uncomfortable pattern emerged: patients whose conditions spanned these traditional specialty boundaries — particularly those with overlapping cardiovascular, renal, and metabolic disease — were faring far worse than the sum of their individually managed conditions would predict. The silo model was not just incomplete. In many cases, it was actively failing the patients it was designed to serve.
The CKM Framework: A New Lens for the Body's Most Interconnected Systems
The formal articulation of the Cardio-Kidney-Metabolic framework represents one of the most significant conceptual advances in internal medicine in recent decades. In 2023, the American Heart Association published a landmark presidential advisory introducing CKM syndrome as an official staging framework — a clinical recognition that cardiovascular disease, chronic kidney disease, and metabolic disorders including obesity and type 2 diabetes are not merely comorbidities that happen to cluster together, but manifestations of a shared, deeply interconnected pathophysiology that must be understood and managed as a unified system. The framework acknowledges what physiologists have long known: the heart, kidneys, liver, adipose tissue, and pancreatic beta cells are in constant bidirectional communication through shared hormonal, hemodynamic, and inflammatory pathways. Disruption of any one node in this network propagates dysfunction throughout the others. The CKM approach does not simply ask which organ is diseased; it asks how the disease state of each organ is generating, amplifying, or perpetuating dysfunction in the others — and which therapeutic interventions can simultaneously benefit multiple systems. This shift from organ-level thinking to system-level thinking is the intellectual foundation of modern cardiometabolic and cardiorenal medicine.
The Continuous Cycle: How Each System Drives the Others
The power of the CKM model lies in its ability to trace the biological mechanisms by which dysfunction propagates around the triangle. Beginning with the metabolic axis: insulin resistance — the defining metabolic lesion of type 2 diabetes and obesity — produces sustained hyperglycemia and dyslipidemia, both of which damage the glomerular endothelium of the kidney through oxidative stress, advanced glycation end-products, and pro-inflammatory cytokines. The result is diabetic nephropathy, the leading cause of end-stage kidney disease globally. As kidney function declines, however, the injury does not remain contained within the renal system. Chronic kidney disease activates the renin-angiotensin-aldosterone system and the sympathetic nervous system, driving fluid retention, sodium imbalance, and sustained hypertension — a direct, relentless insult to the cardiovascular system. The hypertensive load forces the left ventricle to work against chronically elevated afterload, producing left ventricular hypertrophy and diastolic dysfunction, while accelerating atherosclerosis in the coronary and peripheral vasculature. Simultaneously, the uremic milieu of advanced CKD promotes endothelial dysfunction, accelerated vascular calcification, and a pro-coagulant state that dramatically amplifies cardiovascular risk far beyond what hypertension alone would predict. And from the cardiovascular side, reduced cardiac output impairs renal perfusion, worsening ischemic nephropathy and perpetuating the kidney injury that already exists. This is cardiorenal syndrome — one of the most clinically devastating expressions of the CKM cycle, and one that cannot be interrupted by treating the heart or the kidney alone. The therapeutic revolution that the CKM framework enables is embodied by a new class of agents — SGLT2 inhibitors and GLP-1 receptor agonists — that were initially developed as glucose-lowering drugs but have demonstrated simultaneous, independent benefits across all three nodes of the CKM triangle: reducing cardiovascular events, slowing CKD progression, and improving metabolic parameters. These drugs did not break the cycle by treating one component; they intervened at the shared pathophysiological pathways that the cycle depends upon. They are, in the truest sense, CKM drugs — and their emergence is the clearest clinical proof that the integrated framework is not just conceptually elegant but therapeutically transformative.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis