-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
The Skin as a Battleground: A Structural and Cellular Pathology of Cutaneous Tuberculosis
A Disease That Wears Many Faces
Tuberculosis is most readily imagined as a disease of the lungs — a pulmonary process marked by cough, hemoptysis, and the slow excavation of parenchymal tissue under the relentless pressure of Mycobacterium tuberculosis. That image, while accurate as a portrait of the disease's most common clinical expression, obscures a far broader biological reality. Mycobacterium tuberculosis is a systemic pathogen. Given the right conditions — immunological vulnerability, bacterial burden, or a disruption in anatomical barriers — it is capable of establishing itself in virtually any organ system in the human body, including the organ that is simultaneously the most visible and the most frequently overlooked in the context of tuberculosis: the skin.
Cutaneous tuberculosis is not a rarity, but it is a diagnosis that clinicians are poorly trained to suspect. It is estimated to account for between one and two percent of all extrapulmonary tuberculosis cases, a figure that almost certainly underestimates true prevalence in high-burden settings where diagnostic resources are limited and lesions are frequently attributed to other dermatological conditions. Its presentations range from the ulcerating papules of tuberculosis verrucosa cutis to the soft subcutaneous fluctuance of scrofuloderma to the protean, slowly progressive plaques of lupus vulgaris — a morphological variety so broad that no single clinical description can encompass it.
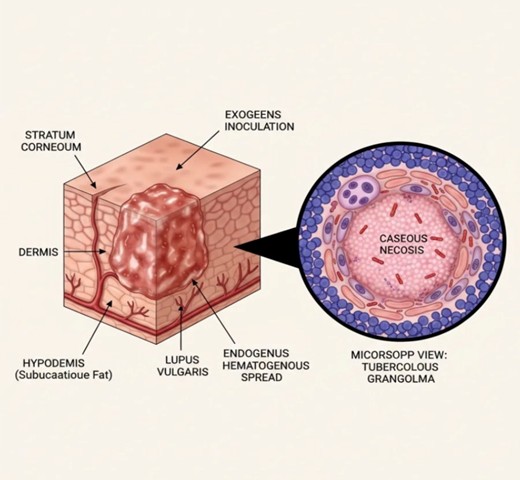
To understand cutaneous tuberculosis — to diagnose it with confidence and treat it with appropriate urgency — requires understanding the disease at two levels simultaneously: the structural level, where routes of bacterial arrival determine the depth and character of the resulting lesion, and the cellular level, where the immune system's attempt to contain the invader leaves a microscopic signature that is, in skilled hands, diagnostic. The figure above presents both perspectives within a single visual framework, and this article traces the pathological logic that connects them.
Cutaneous tuberculosis is not a rarity, but it is a diagnosis that clinicians are poorly trained to suspect. It is estimated to account for between one and two percent of all extrapulmonary tuberculosis cases, a figure that almost certainly underestimates true prevalence in high-burden settings where diagnostic resources are limited and lesions are frequently attributed to other dermatological conditions. Its presentations range from the ulcerating papules of tuberculosis verrucosa cutis to the soft subcutaneous fluctuance of scrofuloderma to the protean, slowly progressive plaques of lupus vulgaris — a morphological variety so broad that no single clinical description can encompass it.
To understand cutaneous tuberculosis — to diagnose it with confidence and treat it with appropriate urgency — requires understanding the disease at two levels simultaneously: the structural level, where routes of bacterial arrival determine the depth and character of the resulting lesion, and the cellular level, where the immune system's attempt to contain the invader leaves a microscopic signature that is, in skilled hands, diagnostic. The figure above presents both perspectives within a single visual framework, and this article traces the pathological logic that connects them.
The Architecture of the Skin: Why It Both Resists and Succumbs
The skin is the human body's primary physical interface with the external environment, and its layered architecture reflects the evolutionary pressure of that role. The outermost layer, the stratum corneum, is a compact lattice of anucleate corneocytes embedded in a lipid-rich extracellular matrix — a barrier of remarkable mechanical integrity that resists microbial penetration under normal circumstances. Beneath it, the viable epidermis provides immunological surveillance through resident Langerhans cells and keratinocyte-derived cytokines. The dermis, rich in fibroblasts, blood vessels, lymphatics, and resident macrophages, constitutes the structural scaffolding of the skin and the primary anatomical territory within which tuberculous lesions develop. The hypodermis, composed predominantly of adipose tissue and larger vascular structures, represents the deepest zone accessible to bacterial spread from the dermis.
This layered organization is not incidental to the pathology of cutaneous tuberculosis — it is central to it. The route by which mycobacteria enter the skin determines which layer is first colonized, how rapidly the infection spreads to adjacent structures, what clinical morphology the resulting lesion assumes, and, ultimately, what diagnostic and therapeutic approach is most appropriate. Two routes are recognized, and their distinction carries clinical consequence.
This layered organization is not incidental to the pathology of cutaneous tuberculosis — it is central to it. The route by which mycobacteria enter the skin determines which layer is first colonized, how rapidly the infection spreads to adjacent structures, what clinical morphology the resulting lesion assumes, and, ultimately, what diagnostic and therapeutic approach is most appropriate. Two routes are recognized, and their distinction carries clinical consequence.
Routes of Entry: How Mycobacterium Tuberculosis Reaches the Skin
Exogenous Inoculation: The Barrier Breached from Without
The first and more intuitively comprehensible route is exogenous inoculation — the direct introduction of mycobacteria into the skin through a breach in the stratum corneum. This barrier breach may be traumatic, arising from an abrasion, laceration, puncture wound, or surgical incision. It may be occupational, as in the historically documented cases of primary inoculation tuberculosis in pathologists, anatomists, and healthcare workers performing procedures on patients with active pulmonary disease without adequate skin protection. It may also occur through tattooing needles, body-piercing instruments, or any percutaneous procedure performed with contaminated equipment.
Once through the stratum corneum, the inoculated mycobacteria encounter the immunological machinery of the dermis. In an immunocompetent host with no prior sensitization to mycobacterial antigens — the tuberculin-naive individual — the initial response is the formation of a primary complex: a slowly enlarging papule or nodule at the inoculation site, accompanied by regional lymphadenopathy. This presentation, historically called the tuberculosis primary complex of the skin or the tuberculous chancre, mirrors in miniature the Ghon complex of primary pulmonary tuberculosis. In sensitized individuals, the response is more brisk, more inflammatory, and more characteristically granulomatous — reflecting immunological memory that accelerates but also partially contains the host response.
The clinical forms of cutaneous tuberculosis arising from exogenous inoculation include tuberculosis verrucosa cutis — a warty, hyperkeratotic plaque that develops at sites of repeated low-level inoculation in sensitized individuals — and, in specific occupational contexts, the primary inoculation complex. Both forms are notable for their superficial or mid-dermal localization and their relative confinement to the anatomical zone of initial bacterial entry.
Endogenous Hematogenous Spread: The Enemy Within
The second route — endogenous hematogenous spread — is both more clinically significant and more diagnostically elusive. Here, the skin is not a primary site of infection but a secondary one: the bacteria originate in an internal focus, most commonly the lungs, mediastinal lymph nodes, or another visceral organ, and reach the dermis by traveling through the bloodstream. The dermal vasculature, particularly its post-capillary venular plexus, serves as the anatomical gateway through which circulating mycobacteria exit the vascular compartment and seed the connective tissue of the dermis.
The clinical implication of hematogenous seeding is profound: it means that a cutaneous lesion attributed to tuberculosis may be the most visible manifestation of a disseminated or reactivation tuberculous process involving organs that are not clinically apparent. The patient who presents with a slowly evolving plaque on the face, neck, or extremity may harbor active pulmonary tuberculosis, tuberculous lymphadenitis, or skeletal tuberculosis that has not yet declared itself through respiratory or systemic symptoms. Recognizing the skin lesion as an endogenous process therefore mandates a comprehensive systemic evaluation — chest imaging, sputum evaluation, tuberculin skin testing or interferon-gamma release assay, and in many cases cross-sectional imaging of the chest and abdomen.
A further and clinically important variant of endogenous spread is contiguous extension — the direct invasion of overlying skin by a subcutaneous tuberculous focus, most characteristically a caseous lymph node. This mechanism underlies scrofuloderma, in which a tuberculous cervical lymph node softens, liquefies, and ultimately ruptures through the skin surface, creating a characteristic collar-stud abscess with a chronic discharging sinus tract. The pathophysiology is not hematogenous in the strict sense, but it is equally endogenous: the source is internal, and the skin is a passive victim of bacterial invasion from below.
The first and more intuitively comprehensible route is exogenous inoculation — the direct introduction of mycobacteria into the skin through a breach in the stratum corneum. This barrier breach may be traumatic, arising from an abrasion, laceration, puncture wound, or surgical incision. It may be occupational, as in the historically documented cases of primary inoculation tuberculosis in pathologists, anatomists, and healthcare workers performing procedures on patients with active pulmonary disease without adequate skin protection. It may also occur through tattooing needles, body-piercing instruments, or any percutaneous procedure performed with contaminated equipment.
Once through the stratum corneum, the inoculated mycobacteria encounter the immunological machinery of the dermis. In an immunocompetent host with no prior sensitization to mycobacterial antigens — the tuberculin-naive individual — the initial response is the formation of a primary complex: a slowly enlarging papule or nodule at the inoculation site, accompanied by regional lymphadenopathy. This presentation, historically called the tuberculosis primary complex of the skin or the tuberculous chancre, mirrors in miniature the Ghon complex of primary pulmonary tuberculosis. In sensitized individuals, the response is more brisk, more inflammatory, and more characteristically granulomatous — reflecting immunological memory that accelerates but also partially contains the host response.
The clinical forms of cutaneous tuberculosis arising from exogenous inoculation include tuberculosis verrucosa cutis — a warty, hyperkeratotic plaque that develops at sites of repeated low-level inoculation in sensitized individuals — and, in specific occupational contexts, the primary inoculation complex. Both forms are notable for their superficial or mid-dermal localization and their relative confinement to the anatomical zone of initial bacterial entry.
Endogenous Hematogenous Spread: The Enemy Within
The second route — endogenous hematogenous spread — is both more clinically significant and more diagnostically elusive. Here, the skin is not a primary site of infection but a secondary one: the bacteria originate in an internal focus, most commonly the lungs, mediastinal lymph nodes, or another visceral organ, and reach the dermis by traveling through the bloodstream. The dermal vasculature, particularly its post-capillary venular plexus, serves as the anatomical gateway through which circulating mycobacteria exit the vascular compartment and seed the connective tissue of the dermis.
The clinical implication of hematogenous seeding is profound: it means that a cutaneous lesion attributed to tuberculosis may be the most visible manifestation of a disseminated or reactivation tuberculous process involving organs that are not clinically apparent. The patient who presents with a slowly evolving plaque on the face, neck, or extremity may harbor active pulmonary tuberculosis, tuberculous lymphadenitis, or skeletal tuberculosis that has not yet declared itself through respiratory or systemic symptoms. Recognizing the skin lesion as an endogenous process therefore mandates a comprehensive systemic evaluation — chest imaging, sputum evaluation, tuberculin skin testing or interferon-gamma release assay, and in many cases cross-sectional imaging of the chest and abdomen.
A further and clinically important variant of endogenous spread is contiguous extension — the direct invasion of overlying skin by a subcutaneous tuberculous focus, most characteristically a caseous lymph node. This mechanism underlies scrofuloderma, in which a tuberculous cervical lymph node softens, liquefies, and ultimately ruptures through the skin surface, creating a characteristic collar-stud abscess with a chronic discharging sinus tract. The pathophysiology is not hematogenous in the strict sense, but it is equally endogenous: the source is internal, and the skin is a passive victim of bacterial invasion from below.
Lupus Vulgaris: The Archetypal Cutaneous Tuberculosis Lesion
Among all the morphological forms of cutaneous tuberculosis, lupus vulgaris occupies a special position — as the most common, the most instructive, and historically the most studied. The term, coined in the nineteenth century to describe its wolfish, destructive quality, refers to a chronic, progressive, pauci-bacillary form of cutaneous tuberculosis that arises most commonly through endogenous spread in individuals with moderate-to-high cellular immunity against mycobacteria. The relative immunocompetence of these patients is, paradoxically, what creates the characteristic lesion morphology: the immune response is vigorous enough to constrain bacterial spread and prevent dissemination, but not sufficient to eradicate the organism.
Lupus vulgaris begins as a small, reddish-brown papule that, on diascopy — the application of a glass slide to the skin surface to blanch erythema — reveals the characteristic apple-jelly color of granulomatous inflammation, a finding so pathognomonic that it has been used clinically for well over a century. Over months to years, the lesion expands centrifugally, developing an irregular, geographic border with active erythematous edges and a central zone that may heal with atrophic scarring, undergo ulceration, or develop the verrucous hyperkeratotic surface texture that reflects chronic epithelial injury and regeneration.
The structural depth of the lupus vulgaris lesion is a critical pathological feature that the figure illustrates with anatomical clarity. The infection is not a surface phenomenon confined to the epidermis. It takes root in the dermis, where the bulk of the granulomatous inflammation is established, and extends downward into the hypodermis — the subcutaneous fat — particularly in long-standing or inadequately treated cases. This depth of invasion has several clinical consequences. It accounts for the indurated, infiltrated quality palpable on clinical examination. It explains why superficial treatment — topical agents, shallow destructive procedures — is wholly inadequate for this disease. And it creates the substrate for the chronic tissue destruction and scarring that, over years of untreated disease, can lead to disfiguring contractures, ectropion, and, in the most severe and prolonged cases, squamous cell carcinoma arising within the field of chronic granulomatous inflammation.
Lupus vulgaris begins as a small, reddish-brown papule that, on diascopy — the application of a glass slide to the skin surface to blanch erythema — reveals the characteristic apple-jelly color of granulomatous inflammation, a finding so pathognomonic that it has been used clinically for well over a century. Over months to years, the lesion expands centrifugally, developing an irregular, geographic border with active erythematous edges and a central zone that may heal with atrophic scarring, undergo ulceration, or develop the verrucous hyperkeratotic surface texture that reflects chronic epithelial injury and regeneration.
The structural depth of the lupus vulgaris lesion is a critical pathological feature that the figure illustrates with anatomical clarity. The infection is not a surface phenomenon confined to the epidermis. It takes root in the dermis, where the bulk of the granulomatous inflammation is established, and extends downward into the hypodermis — the subcutaneous fat — particularly in long-standing or inadequately treated cases. This depth of invasion has several clinical consequences. It accounts for the indurated, infiltrated quality palpable on clinical examination. It explains why superficial treatment — topical agents, shallow destructive procedures — is wholly inadequate for this disease. And it creates the substrate for the chronic tissue destruction and scarring that, over years of untreated disease, can lead to disfiguring contractures, ectropion, and, in the most severe and prolonged cases, squamous cell carcinoma arising within the field of chronic granulomatous inflammation.
The Tuberculous Granuloma: The Immune System's Fortress and Its Flaw
Formation and Structure
At the cellular level, the hallmark of tuberculosis — in the lung, the lymph node, the bone, or the skin — is the granuloma. This structure is not a passive collection of inflammatory cells but a highly organized, dynamically maintained immunological fortress built and maintained by a coordinated response involving macrophages, T lymphocytes, B lymphocytes, dendritic cells, fibroblasts, and the cytokine networks that bind them together. Understanding the granuloma is understanding tuberculosis, because the granuloma is simultaneously the body's most effective mechanism for containing mycobacterial spread and the structure whose failure leads to progressive disease.
The genesis of the tuberculous granuloma begins with the phagocytosis of mycobacteria by tissue-resident macrophages. Unlike pyogenic bacteria, which are rapidly destroyed within the phagolysosomal compartment, Mycobacterium tuberculosis has evolved an array of virulence mechanisms that allow it to survive and replicate within the macrophage: inhibition of phagosome-lysosome fusion, resistance to reactive oxygen and nitrogen intermediates, and modulation of the macrophage's own apoptotic signaling to prevent the cell death that would otherwise limit bacterial spread.
As infected macrophages present mycobacterial antigens to T lymphocytes and receive activating signals in return, they differentiate into epithelioid cells — large, pale, polygonally shaped cells with abundant eosinophilic cytoplasm and a characteristic shoe-shaped nucleus, adapted for secretion of pro-inflammatory mediators rather than the phagocytic function of their precursors. Multiple epithelioid cells fuse under the influence of interferon-gamma and TNF-alpha to form Langhans giant cells — the multinucleated, peripherally ring-arranged-nuclei giant cells that are among the most recognizable histological features of the tuberculous granuloma and a defining criterion in the pathological diagnosis of tuberculosis.
The mature granuloma, as illustrated in the microscopic callout of Figure 1, is organized in concentric zones. At its outer rim, a dense mantle of lymphocytes — predominantly CD4-positive T helper cells — provides continuous immunological surveillance and cytokine support for the cells within. Interior to this lymphocytic cuff lies the zone of epithelioid cells and Langhans giant cells, the primary cellular machinery of mycobacterial containment. And at the very center lies the structure that gives the tuberculous granuloma its defining and diagnostically decisive character: the zone of caseous necrosis.
Caseous Necrosis: The Center That Cannot Hold
Caseous necrosis — named for its gross resemblance to soft white cheese — is a form of cell death that is both the product of intense immunological activity and the signal of its partial failure. It arises through the combined effects of direct mycobacterial cytotoxicity, the elaboration of lytic enzymes by activated macrophages, and a poorly characterized form of programmed necrosis that is distinct from apoptosis and that appears to be specifically triggered by mycobacterial virulence factors. The result is a central zone of amorphous, acellular, eosinophilic material — tissue that has lost its structural identity and histological architecture, reduced to a featureless, necrotic core.
Caseous necrosis is pathologically important for several reasons beyond its diagnostic utility. It creates a microenvironment — anoxic, acidic, nutrient-depleted — that is hostile to both host immune cells and, to a lesser extent, the mycobacteria themselves. Paradoxically, this hostile environment also renders the caseous center partially resistant to the penetration of antituberculous drugs, which depend on metabolically active bacteria for their full bactericidal effect. The necrotic core thus becomes a reservoir from which viable but dormant mycobacteria can persist for prolonged periods, resistant to immune clearance and partially shielded from pharmacological attack.
In the skin, the clinical consequences of caseation are visible as the softening and liquefaction that precede ulceration in lupus vulgaris and as the fluctuant subcutaneous masses of scrofuloderma. When the liquid caseous material breaches the overlying skin surface, it creates the characteristic ulcers, sinus tracts, and discharging lesions that are among the most diagnostically compelling — and most disfiguring — manifestations of cutaneous tuberculosis.
The Granuloma as Diagnostic Signature
The histological identification of the tuberculous granuloma — with its characteristic combination of epithelioid cells, Langhans giant cells, lymphocytic cuffing, and central caseation — remains the cornerstone of the pathological diagnosis of cutaneous tuberculosis. Skin biopsy, with tissue submitted for both histopathology and mycobacterial culture, is the diagnostic procedure of choice whenever cutaneous tuberculosis is clinically suspected. The granulomatous architecture is detectable even in pauci-bacillary lesions where acid-fast staining and culture may yield negative results, making histopathology uniquely valuable in the context of this diagnosis.
It is important to acknowledge that the tuberculous granuloma is not entirely specific to Mycobacterium tuberculosis. Other mycobacterial species, fungal infections, sarcoidosis, foreign body reactions, and certain parasitic infections can produce granulomatous inflammation in the skin that is histologically similar. The distinction between these entities requires clinical correlation, culture results, molecular diagnostic techniques including polymerase chain reaction for mycobacterial DNA, and in some cases immunohistochemical staining. Nevertheless, the combination of epithelioid granulomas with caseation in the appropriate epidemiological and clinical context carries a very high positive predictive value for mycobacterial disease, and the initiation of antituberculous therapy should not necessarily await culture confirmation when the clinical and histological picture is sufficiently compelling.
At the cellular level, the hallmark of tuberculosis — in the lung, the lymph node, the bone, or the skin — is the granuloma. This structure is not a passive collection of inflammatory cells but a highly organized, dynamically maintained immunological fortress built and maintained by a coordinated response involving macrophages, T lymphocytes, B lymphocytes, dendritic cells, fibroblasts, and the cytokine networks that bind them together. Understanding the granuloma is understanding tuberculosis, because the granuloma is simultaneously the body's most effective mechanism for containing mycobacterial spread and the structure whose failure leads to progressive disease.
The genesis of the tuberculous granuloma begins with the phagocytosis of mycobacteria by tissue-resident macrophages. Unlike pyogenic bacteria, which are rapidly destroyed within the phagolysosomal compartment, Mycobacterium tuberculosis has evolved an array of virulence mechanisms that allow it to survive and replicate within the macrophage: inhibition of phagosome-lysosome fusion, resistance to reactive oxygen and nitrogen intermediates, and modulation of the macrophage's own apoptotic signaling to prevent the cell death that would otherwise limit bacterial spread.
As infected macrophages present mycobacterial antigens to T lymphocytes and receive activating signals in return, they differentiate into epithelioid cells — large, pale, polygonally shaped cells with abundant eosinophilic cytoplasm and a characteristic shoe-shaped nucleus, adapted for secretion of pro-inflammatory mediators rather than the phagocytic function of their precursors. Multiple epithelioid cells fuse under the influence of interferon-gamma and TNF-alpha to form Langhans giant cells — the multinucleated, peripherally ring-arranged-nuclei giant cells that are among the most recognizable histological features of the tuberculous granuloma and a defining criterion in the pathological diagnosis of tuberculosis.
The mature granuloma, as illustrated in the microscopic callout of Figure 1, is organized in concentric zones. At its outer rim, a dense mantle of lymphocytes — predominantly CD4-positive T helper cells — provides continuous immunological surveillance and cytokine support for the cells within. Interior to this lymphocytic cuff lies the zone of epithelioid cells and Langhans giant cells, the primary cellular machinery of mycobacterial containment. And at the very center lies the structure that gives the tuberculous granuloma its defining and diagnostically decisive character: the zone of caseous necrosis.
Caseous Necrosis: The Center That Cannot Hold
Caseous necrosis — named for its gross resemblance to soft white cheese — is a form of cell death that is both the product of intense immunological activity and the signal of its partial failure. It arises through the combined effects of direct mycobacterial cytotoxicity, the elaboration of lytic enzymes by activated macrophages, and a poorly characterized form of programmed necrosis that is distinct from apoptosis and that appears to be specifically triggered by mycobacterial virulence factors. The result is a central zone of amorphous, acellular, eosinophilic material — tissue that has lost its structural identity and histological architecture, reduced to a featureless, necrotic core.
Caseous necrosis is pathologically important for several reasons beyond its diagnostic utility. It creates a microenvironment — anoxic, acidic, nutrient-depleted — that is hostile to both host immune cells and, to a lesser extent, the mycobacteria themselves. Paradoxically, this hostile environment also renders the caseous center partially resistant to the penetration of antituberculous drugs, which depend on metabolically active bacteria for their full bactericidal effect. The necrotic core thus becomes a reservoir from which viable but dormant mycobacteria can persist for prolonged periods, resistant to immune clearance and partially shielded from pharmacological attack.
In the skin, the clinical consequences of caseation are visible as the softening and liquefaction that precede ulceration in lupus vulgaris and as the fluctuant subcutaneous masses of scrofuloderma. When the liquid caseous material breaches the overlying skin surface, it creates the characteristic ulcers, sinus tracts, and discharging lesions that are among the most diagnostically compelling — and most disfiguring — manifestations of cutaneous tuberculosis.
The Granuloma as Diagnostic Signature
The histological identification of the tuberculous granuloma — with its characteristic combination of epithelioid cells, Langhans giant cells, lymphocytic cuffing, and central caseation — remains the cornerstone of the pathological diagnosis of cutaneous tuberculosis. Skin biopsy, with tissue submitted for both histopathology and mycobacterial culture, is the diagnostic procedure of choice whenever cutaneous tuberculosis is clinically suspected. The granulomatous architecture is detectable even in pauci-bacillary lesions where acid-fast staining and culture may yield negative results, making histopathology uniquely valuable in the context of this diagnosis.
It is important to acknowledge that the tuberculous granuloma is not entirely specific to Mycobacterium tuberculosis. Other mycobacterial species, fungal infections, sarcoidosis, foreign body reactions, and certain parasitic infections can produce granulomatous inflammation in the skin that is histologically similar. The distinction between these entities requires clinical correlation, culture results, molecular diagnostic techniques including polymerase chain reaction for mycobacterial DNA, and in some cases immunohistochemical staining. Nevertheless, the combination of epithelioid granulomas with caseation in the appropriate epidemiological and clinical context carries a very high positive predictive value for mycobacterial disease, and the initiation of antituberculous therapy should not necessarily await culture confirmation when the clinical and histological picture is sufficiently compelling.
Clinical Diagnosis: Connecting the Structural and Cellular to the Bedside
The diagnostic challenge of cutaneous tuberculosis lies not in the complexity of the pathological findings — which, when present, are highly instructive — but in the clinical threshold for suspecting the diagnosis in the first place. Lupus vulgaris is misdiagnosed as psoriasis, discoid lupus erythematosus, sarcoidosis, or chronic fungal infection. Scrofuloderma is attributed to atypical mycobacteria, actinomycosis, or infected sebaceous cysts. Tuberculosis verrucosa cutis is confused with viral warts, chromoblastomycosis, or hypertrophic lichen planus. The morphological diversity of cutaneous tuberculosis, combined with its relative rarity in low-burden settings, means that it frequently spends months or years on a differential diagnosis list without reaching the top.
The clinician who understands the pathological framework described in this article — who recognizes that a chronic, indurated, slowly progressive plaque extending deep into the dermis and hypodermis may represent an endogenous hematogenous seeding from an internal tuberculous focus, and who connects that macroscopic observation to the microscopic granulomatous architecture that awaits discovery on biopsy — is far better equipped to make this diagnosis at a clinically actionable stage than one who relies on morphological pattern recognition alone.
The diagnostic workup of suspected cutaneous tuberculosis should be systematic and comprehensive. Punch or incisional biopsy from the active edge of the lesion — not the central necrotic zone, where viable tissue is absent — should be submitted in formalin for histopathology and in saline or mycobacterial transport medium for culture and polymerase chain reaction. Systemic evaluation for concurrent active tuberculosis should include chest radiography, and in high-clinical-suspicion cases, computed tomography of the thorax and abdomen. Immunological testing with tuberculin skin test or interferon-gamma release assay supports the diagnosis but cannot distinguish active from latent infection. HIV testing is mandatory, given the dramatically altered clinical presentation, tissue response, and prognosis of cutaneous tuberculosis in the setting of advanced immunosuppression.
The clinician who understands the pathological framework described in this article — who recognizes that a chronic, indurated, slowly progressive plaque extending deep into the dermis and hypodermis may represent an endogenous hematogenous seeding from an internal tuberculous focus, and who connects that macroscopic observation to the microscopic granulomatous architecture that awaits discovery on biopsy — is far better equipped to make this diagnosis at a clinically actionable stage than one who relies on morphological pattern recognition alone.
The diagnostic workup of suspected cutaneous tuberculosis should be systematic and comprehensive. Punch or incisional biopsy from the active edge of the lesion — not the central necrotic zone, where viable tissue is absent — should be submitted in formalin for histopathology and in saline or mycobacterial transport medium for culture and polymerase chain reaction. Systemic evaluation for concurrent active tuberculosis should include chest radiography, and in high-clinical-suspicion cases, computed tomography of the thorax and abdomen. Immunological testing with tuberculin skin test or interferon-gamma release assay supports the diagnosis but cannot distinguish active from latent infection. HIV testing is mandatory, given the dramatically altered clinical presentation, tissue response, and prognosis of cutaneous tuberculosis in the setting of advanced immunosuppression.
Treatment: The Same Regimen, a Different Stage
The treatment of cutaneous tuberculosis does not differ in principle from the treatment of pulmonary or other forms of extrapulmonary tuberculosis. Standard first-line antituberculous therapy — the combination of isoniazid, rifampicin, pyrazinamide, and ethambutol for an initial intensive phase of two months, followed by isoniazid and rifampicin for a continuation phase of four to seven months — is the backbone of management for all drug-susceptible cutaneous tuberculosis forms. Drug susceptibility testing should guide therapy whenever possible, particularly in regions or patient populations with elevated rates of drug resistance.
What distinguishes the management of cutaneous tuberculosis from its pulmonary counterpart is not the pharmacology but the monitoring: lesion response — regression of induration, reduction in lesion area, re-epithelialization of ulcers — provides a visible and directly measurable endpoint that is not available in pulmonary disease. Clinical photography at the initiation of therapy and at regular intervals serves both as a monitoring tool and as a medicolegal record. Failure to demonstrate progressive improvement within the first six to eight weeks of therapy should prompt reassessment of the diagnosis, consideration of drug-resistant disease, and review of adherence.
The role of adjunctive surgery is limited but not absent. Scrofuloderma with large fluctuant masses may benefit from incision and drainage to reduce the volume of necrotic material and accelerate healing, though this should always be performed under antituberculous cover. Longstanding lupus vulgaris lesions that have produced disfiguring contractures or functional impairment may ultimately require reconstructive procedures, but these should be deferred until full microbiological clearance has been achieved.
What distinguishes the management of cutaneous tuberculosis from its pulmonary counterpart is not the pharmacology but the monitoring: lesion response — regression of induration, reduction in lesion area, re-epithelialization of ulcers — provides a visible and directly measurable endpoint that is not available in pulmonary disease. Clinical photography at the initiation of therapy and at regular intervals serves both as a monitoring tool and as a medicolegal record. Failure to demonstrate progressive improvement within the first six to eight weeks of therapy should prompt reassessment of the diagnosis, consideration of drug-resistant disease, and review of adherence.
The role of adjunctive surgery is limited but not absent. Scrofuloderma with large fluctuant masses may benefit from incision and drainage to reduce the volume of necrotic material and accelerate healing, though this should always be performed under antituberculous cover. Longstanding lupus vulgaris lesions that have produced disfiguring contractures or functional impairment may ultimately require reconstructive procedures, but these should be deferred until full microbiological clearance has been achieved.
Conclusion: Seeing the Disease at Every Scale
Cutaneous tuberculosis is a disease that rewards the clinician who thinks at multiple scales simultaneously. At the macroscopic scale, it demands an understanding of routes of bacterial entry and the structural layers of the skin that those routes traverse — an understanding that determines what the lesion looks like, where in the skin it is anchored, and what systemic processes may be driving it from within. At the microscopic scale, it reveals a cellular architecture — the tuberculous granuloma with its caseous necrotic core — that is among the most informative and diagnostically powerful histological patterns in all of infectious disease pathology.
The figure that opens this article is not merely an illustration of a disease. It is a pedagogical argument: that the macroscopic and microscopic are not separate stories about cutaneous tuberculosis but the same story told at different magnifications. The lupus vulgaris plaque that a dermatologist examines under a loupe and the granuloma that a pathologist examines under a microscope are the same pathological event — the same bacterial invasion, the same immunological response, the same dynamic tension between containment and progression — simply rendered visible at different scales.
For a disease that has remained globally prevalent across two centuries of medical progress, that kind of integrated understanding is not an academic luxury. It is the perceptual foundation on which earlier diagnosis, more targeted treatment, and ultimately better outcomes for patients with cutaneous tuberculosis must be built.
The figure that opens this article is not merely an illustration of a disease. It is a pedagogical argument: that the macroscopic and microscopic are not separate stories about cutaneous tuberculosis but the same story told at different magnifications. The lupus vulgaris plaque that a dermatologist examines under a loupe and the granuloma that a pathologist examines under a microscope are the same pathological event — the same bacterial invasion, the same immunological response, the same dynamic tension between containment and progression — simply rendered visible at different scales.
For a disease that has remained globally prevalent across two centuries of medical progress, that kind of integrated understanding is not an academic luxury. It is the perceptual foundation on which earlier diagnosis, more targeted treatment, and ultimately better outcomes for patients with cutaneous tuberculosis must be built.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis