-
-
-
Jakarta, Indonesia
The Silent Atrophy: Why We Are Rewriting Rules on Major Depressive Disorders?
When Do We Need Brain and Spine Surgery? Indications for Neurosurgical Intervention
The Surgical Threshold: When Conservative Care Is No Longer Enough
Neurosurgery is not a first resort. It is, by its nature and by the informed consent philosophy that governs its practice, an intervention of last resort — or of urgent necessity. Every patient who arrives at a neurosurgeon's consultation has typically travelled a road that includes imaging, pharmacological management, physiotherapy, and specialist review before the question of surgery is formally placed on the table. The decision to operate on the brain or spine is governed by a precise calculus that weighs the risks inherent to any intracranial or intraspinal procedure — bleeding, infection, neurological deficit, anaesthetic risk, and recovery burden — against the anticipated natural history of the untreated condition. The threshold is generally crossed under three distinct circumstances: first, when a structural lesion is producing progressive neurological deterioration that is outpacing any realistic expectation of spontaneous or pharmacologically mediated recovery; second, when a lesion is exerting mass effect that threatens imminent herniation or catastrophic vascular compromise; and third, when an acute emergency — haemorrhage, acute hydrocephalus, or traumatic compression — demands immediate decompression to preserve life or neurological function. Understanding this threshold is essential not just for the clinician but for the patient, who must comprehend why the timing of intervention carries as much weight as the decision to intervene at all.
The Principal Indications: From Tumors to Trauma
The spectrum of conditions that bring a patient to neurosurgical intervention is broad, but it resolves into recognizable clinical categories each with its own operative logic. Brain tumors — encompassing primary gliomas ranging from low-grade pilocytic astrocytomas to the devastatingly aggressive glioblastoma multiforme, along with extra-axial tumors such as meningiomas, pituitary adenomas, and cerebral metastases from systemic malignancies — represent the largest surgical volume in cranial neurosurgery. The role of surgery in these cases ranges from cytoreductive resection aimed at reducing tumor burden and relieving intracranial hypertension, to stereotactic biopsy for histological diagnosis without open craniotomy, to stereotactic radiosurgery in selected cases where size, location, or patient comorbidity precludes open resection. Spinal pathology is equally diverse and constitutes a large proportion of neurosurgical operative workload: disc herniation with radiculopathy that is failing conservative management, cervical myelopathy from spondylotic cord compression, lumbar spinal stenosis producing neurogenic claudication, and segmental instability requiring arthrodesis are among the most common indications encountered in elective practice. Cerebrovascular conditions — intracranial aneurysms at risk of rupture or already ruptured, arteriovenous malformations, cavernous malformations with recurrent haemorrhage, and spontaneous intracerebral haemorrhage with mass effect — require intervention calibrated to the specific vascular anatomy and rupture risk. Refractory epilepsy, hydrocephalus requiring shunting or endoscopic third ventriculostomy, central nervous system infections such as cerebral abscess, and acute neurotrauma including subdural and extradural haematoma with clinical deterioration complete the principal indications that define the scope of modern neurosurgical practice.
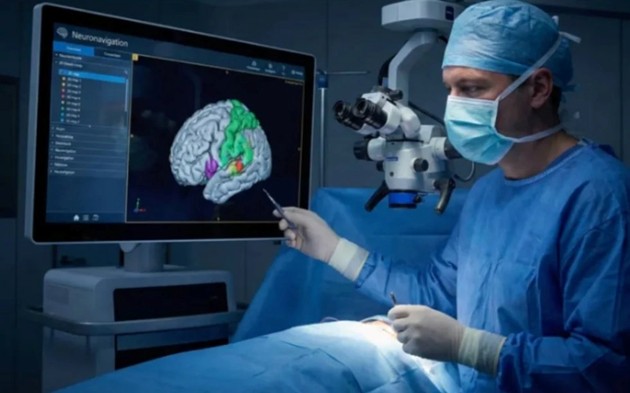
Neuronavigation: The GPS That Guides Every Incision
The revolution that has most profoundly transformed the safety and precision of modern neurosurgery is not a new drug or a new operative technique — it is information. Neuronavigation, the intraoperative application of pre-operative imaging data to provide real-time spatial localization within the operating field, has fundamentally changed the surgeon's relationship with the operative anatomy. Before a craniotomy begins, the patient undergoes high-resolution MRI incorporating structural sequences, diffusion tensor imaging to map white matter tracts, and functional MRI to identify eloquent cortical regions such as the motor cortex, Broca's area, and visual cortex. These datasets are loaded into the neuronavigation workstation and co-registered to the patient's actual head position using fiducial markers or surface-based registration algorithms. The result is a continuously updated three-dimensional map — visible on large intraoperative monitors — that allows the surgeon to know, at any moment during the procedure, precisely where a probe, retractor, or resection cavity lies in relation to critical anatomical structures. This capability is transformative in tumor surgery, where the boundary between neoplastic tissue and functionally vital brain can be millimeters thin, and where the difference between a radical resection that improves survival and a resection that leaves the patient hemiplegic or aphasic may come down to a single errant passage through eloquent cortex. When combined with intraoperative neurophysiological monitoring — which provides real-time electrical feedback from the motor and sensory pathways being approached — neuronavigation creates a safety envelope around the operative field that was simply unavailable to the previous generation of neurosurgeons.
Microscopic Precision: The Surgeon, the Microscope, and the Millimeter That Matters
If neuronavigation provides the map, the surgical microscope provides the lens through which the surgeon reads and acts upon it. The operating microscope — introduced into neurosurgery by the pioneering work of Theodore Kurze and Gazi Yasargil in the 1960s — is not merely a magnifying device; it is the instrument that made microsurgery of the nervous system conceptually and technically possible. Modern neurosurgical microscopes offer variable magnification up to 40 times, coaxial illumination that floods the deepest surgical corridors with high-quality light without casting shadows, and integration with neuronavigation overlays that superimpose navigated anatomy directly into the surgeon's ocular field. Under the microscope, the neurosurgeon works in a visual world entirely different from the naked-eye operative field: cranial nerves, perforating arteries measuring fractions of a millimeter, and the fine architecture of the arachnoid cisterns are visible and manipulable with instruments designed to match this scale — micro-scissors, jeweler's forceps, and bipolar coagulators with tip gaps measured in tenths of a millimeter. The combination of neuronavigation-guided spatial awareness with microsurgical visual precision and the real-time neurophysiological feedback of intraoperative monitoring represents the current pinnacle of neurosurgical technology — a system in which every incision is informed, every trajectory is planned, and the biological cost of accessing the lesion is minimized to the greatest extent that modern science permits. For the patient, this translates not just into technical operative success, but into preservation of the neurological function that defines the quality of the life that surgery is meant to protect.
Motion is Lotion: Why the Gym is the Unexpected Best Friend of Your Osteoarthritis
Reframing physical activity from a feared aggravator to the most evidence-based, non-surgical intervention available for joint pain, stiffness, and functional decline in osteoarthritis